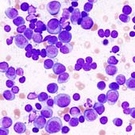
A recent study demonstrated that it is possible to identify multiple myeloma patients with a poor prognosis, based on the presence of rapidly dividing, or proliferating, cancerous cells. Classifying rapidly dividing myeloma cells as a prognostic factor may help patients receive more targeted treatment, especially as anti-proliferative drugs become available.
“Proliferation has long been known to be an adverse prognostic factor in myeloma, but it is quite laborious to measure it in the laboratory, and is thus not routinely measured …
Read the full story »

Patients with monoclonal gammopathy of undetermined significance have an increased risk of developing blood clots, according to a recent study published in the European Journal of Haematology. Blood clots were also associated with poor survival in these patients.
“Our findings underline the risk of VTE [blood cots] in the monoclonal gammopathies in general and might in this way serve to increase the awareness of this complication in myeloma patients,” stated Dr. Henrik Gregersen, of the Aalborg Hospital in Denmark and …
Read the full story »

A 30 mg monthly dose of the bisphosphonate Aredia prevents bone disease as effectively as a 90 mg dose in newly diagnosed multiple myeloma patients, according to a recent Nordic study. The study also found that the lower dose resulted in fewer side effects than the higher dose.
Based on their findings, the researchers recommended 30 mg Aredia be administered to multiple myeloma patients for the prevention of bone disease.
“I believe that our study can lead to a reduction in …
Read the full story »

The results of a recent Phase 2 clinical trial indicate that the new drug Istodax, administered alone, is unlikely to achieve a significant response rate in therapy-resistant multiple myeloma patients. However, the results indicate that that Istodax may help stabilize M-protein production, resolve high calcium levels, or reduce bone pain in some multiple myeloma patients.
Istodax (romidepsin) is a cyclic peptide that inhibits an enzyme in cancer cells known as histone deacetylase (HDAC). By inhibiting HDAC, Istodax disrupts the cell …
Read the full story »
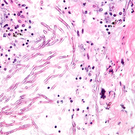
A small study in Japan showed that relapsed multiple myeloma patients treated simultaneously with Velcade and itraconazole experienced greater Velcade-induced side effects, such as lower platelet levels and nerve damage to the extremities, than patients who did not receive itraconazole.
The researchers recommended that multiple myeloma patients receiving both Velcade (bortezomib) and itraconazole (Sporanox) be closely monitored for Velcade-induced side effects throughout the duration of their treatment.
However, according to Dr. Paul Richardson of the Dana-Farber Cancer Institute in …
Read the full story »

ArthroCare Corp. has received clearance from the U.S. Food and Drug Administration to market its Parallax Contour Vertebral Augmentation Device. The Contour device can now be used during minimally invasive surgical procedures to treat spinal fractures caused by multiple myeloma bone lesions.
Weakening of the vertebrae, the bones that make up the spine, can result in vertebral compression fractures. Though the most common cause of vertebral compression fractures is osteoporosis, they may also be caused by multiple myeloma tumors.
Patients …
Read the full story »
| Brand Name: | |
| Generic Name: | |
| Code Name: | BT-062 |
| Company: | Biotest |
| FDA Clinical Phase: | 1/2 |
Description:
BT-062 (news articles) is a toxic drug bound to an antibody that helps deliver the treatment to myeloma and other cancer cells. When …
Read the full story »
